0927HW
20220927
•Use Spartan or WebMO to calculate the ionization energy of atoms with atomic number 1~10, by using CCSD(T)、MP4 and MP2 theoretical method with 6-31G* and 6-311+G(2df,2p) basis set, and do some simple statistical analysis. The unit of the ionization energy is kcal/mol .
•Organize the calculated data of ionization energy, and compare it with the experimental value, then present the result on your exercise page.
- Ionization energy
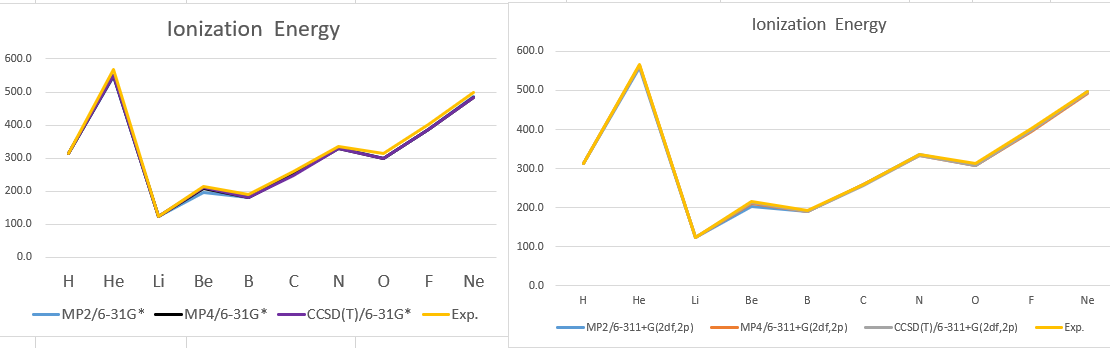
| MP2/6-31G* | MP4/6-31G* | CCSD(T)/6-31G* | exp | |
| H | 312.6 | 312.6 | 312.6 | 313.6 |
| He | 547.7 | 549.9 | 550.0 | 567.3 |
| Li | 122.9 | 122.9 | 122.9 | 124.3 |
| Be | 197.5 | 207.1 | 210.0 | 214.9 |
| B | 182.4 | 180.8 | 179.8 | 191.2 |
| C | 252.2 | 250.2 | 249.0 | 259.6 |
| N | 332.6 | 330.3 | 328.9 | 335.1 |
| O | 298.5 | 298.9 | 298.8 | 313.9 |
| F | 387.6 | 386.4 | 386.2 | 401.7 |
| Ne | 487.4 | 484.3 | 483.8 | 497.2 |
ref :https://physics.nist.gov/PhysRefData/ASD/ionEnergy.html unit:kcal/mol
| MP2/6-311G(2df,2p) | MP4/6-311G(2df,2p) | CCSD(T)/6-311G(2df,2p) | exp | |
| H | 313.6 | 313.6 | 313.6 | 313.6 |
| He | 559.2 | 563.2 | 563.3 | 567.3 |
| Li | 123.1 | 123.1 | 123.1 | 124.3 |
| Be | 203.0 | 211.1 | 214.1 | 214.9 |
| B | 190.2 | 189.6 | 188.9 | 191.2 |
| C | 259.3 | 258.4 | 257.6 | 259.6 |
| N | 336.4 | 334.4 | 333.6 | 335.1 |
| O | 306.7 | 307.4 | 308.3 | 313.9 |
| F | 398.2 | 395.9 | 396.9 | 401.7 |
| Ne | 498.5 | 492.9 | 493.6 | 497.2 |
unit:kcal/mol
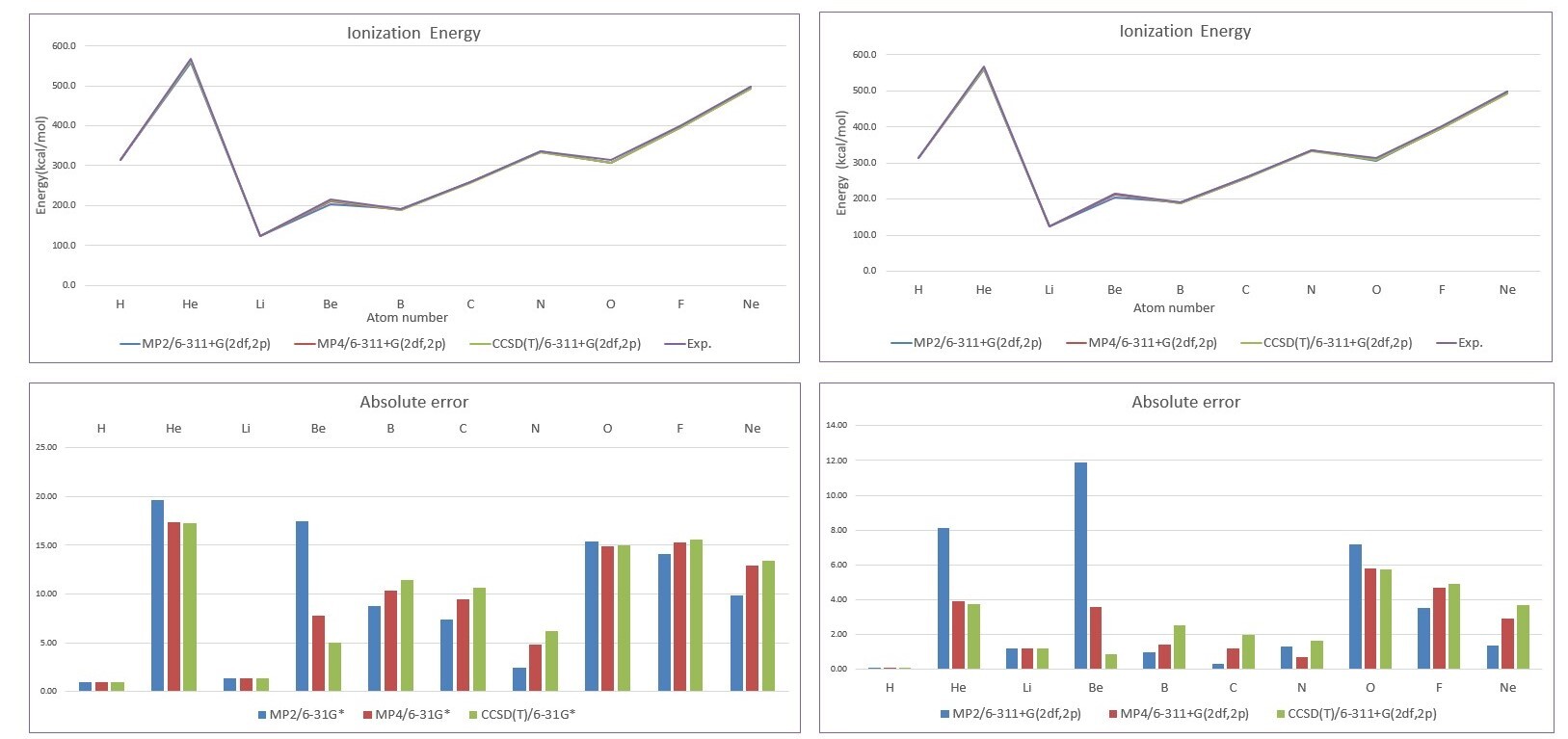
- Absolute error
| MP2/6-31G* | MP4/6-31G* | CCSD(T)/6-31G* | MP2/6-311G(2df,2p) | MP4/6-311G(2df,2p) | CCSD(T)/6-311G(2df,2p) | |
| H | 0.9 | 0.9 | 0.9 | 0.1 | 0.1 | 0.1 |
| He | 19.6 | 17.4 | 17.3 | 8.1 | 3.9 | 3.7 |
| Li | 1.4 | 1.3 | 1.3 | 1.2 | 1.2 | 1.2 |
| Be | 17.5 | 7.8 | 5.0 | 11.9 | 3.5 | 0.9 |
| B | 8.8 | 10.3 | 11.4 | 0.9 | 1.4 | 2.5 |
| C | 7.4 | 9.4 | 10.6 | 0.3 | 1.2 | 2.0 |
| N | 2.5 | 4.8 | 6.2 | 1.3 | 0.7 | 1.6 |
| O | 15.4 | 14.9 | 15.0 | 7.2 | 5.8 | 5.7 |
| F | 14.1 | 15.3 | 15.5 | 3.5 | 4.7 | 4.9 |
| Ne | 9.8 | 12.9 | 13.4 | 1.3 | 2.9 | 3.7 |
| MUE | 9.7 | 9.5 | 9.6 | 3.6 | 2.5 | 2.6 |
- Analysis
Conclusions
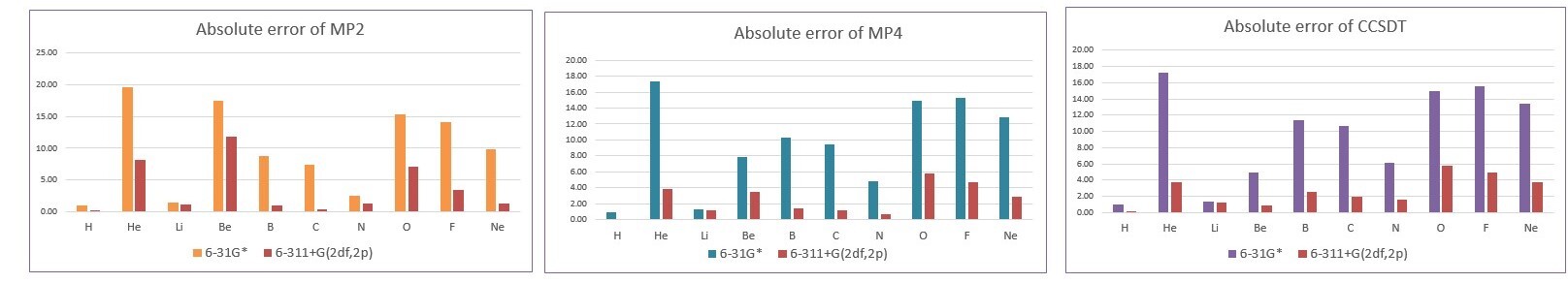
- 相同基底 6-31G*,不同方法的MUE為:MP2 (9.7 kcal/mol) > CCSDT (9.6 kcal/mol) > MP4 (9.5 kcal/mol)
- 相同基底 6-311+G(2df,2p),不同方法的MUE為:MP2 (3.6 kcal/mol) > CCSDT (2.6 kcal/mol) > MP4 (2.5 kcal/mol)
- 利用6-311+G(2df,2p)基底函數誤差比6-31G*小
- MP4方法較準確
