
1.Find the transition state and calculate the barrier height for CH3F + F− → CH3F + F− with following model.
Model: Gas/Microsolvation/Continuum Model ( PCM,sovlent=Water ) Method: MP2/aug-cc-pVDZ
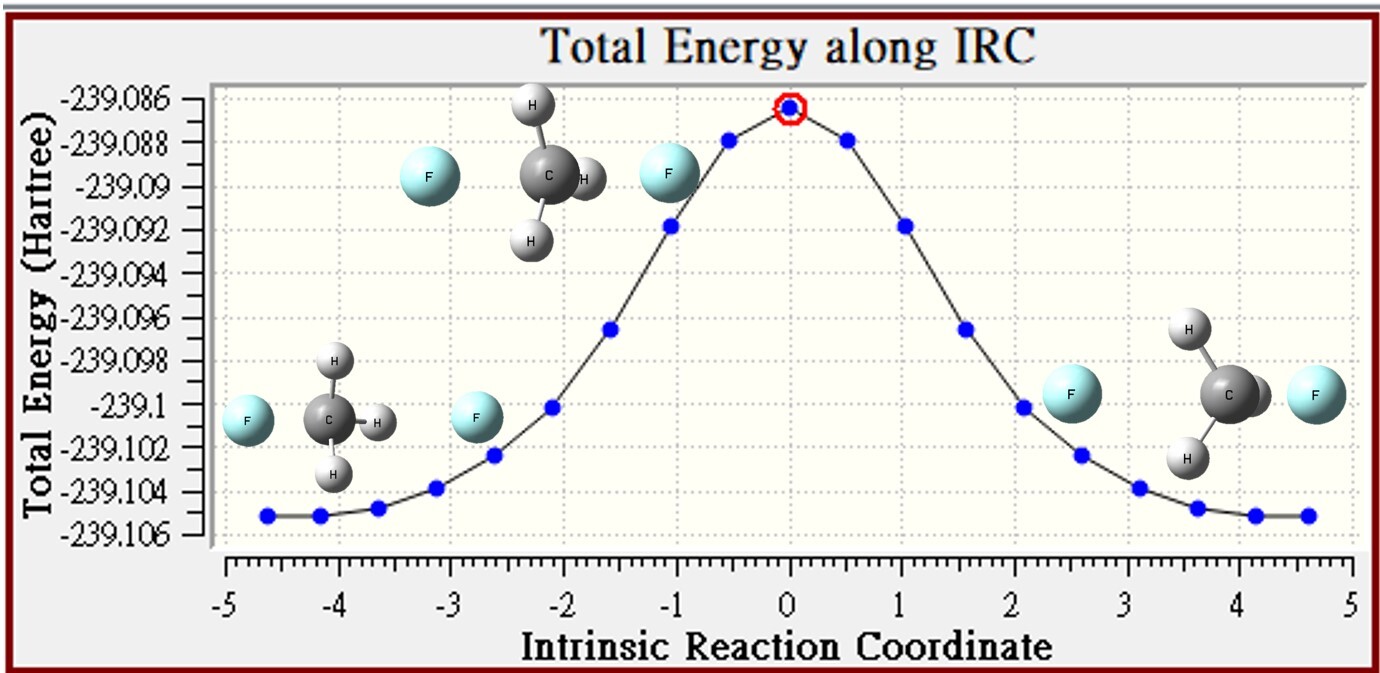
1.Gas
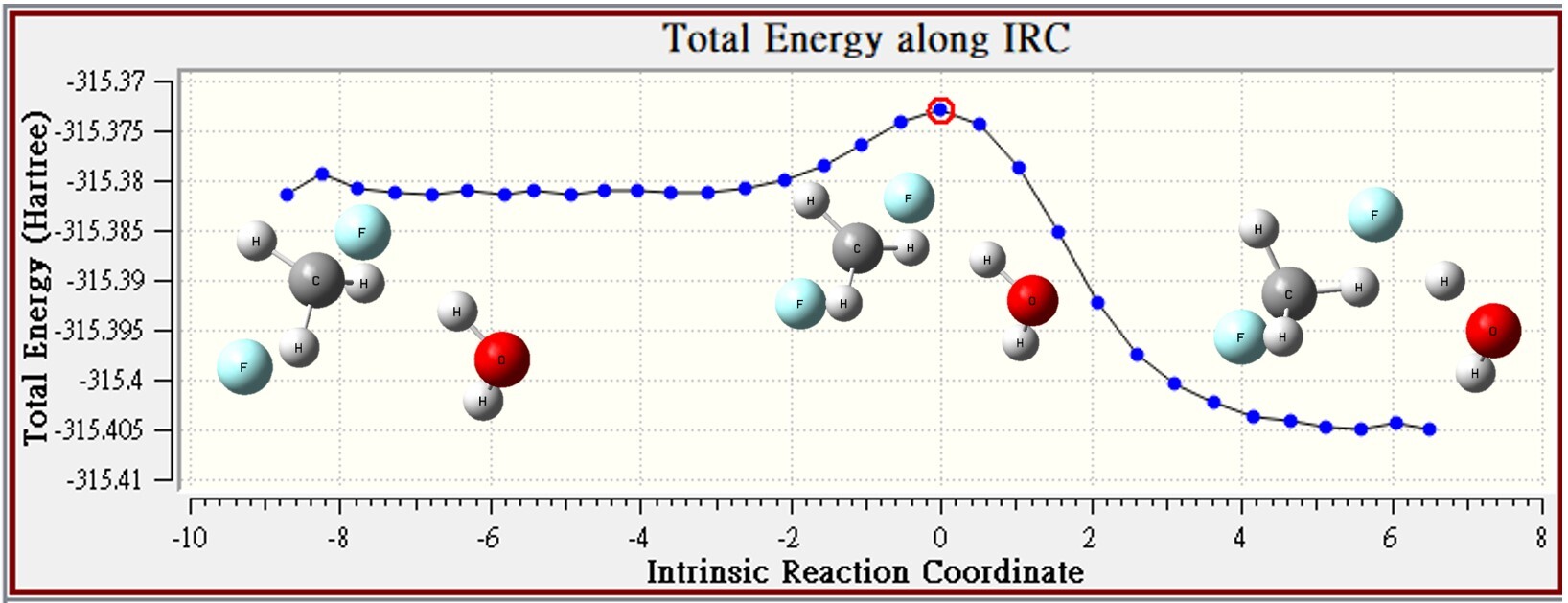
2.Microsolvation
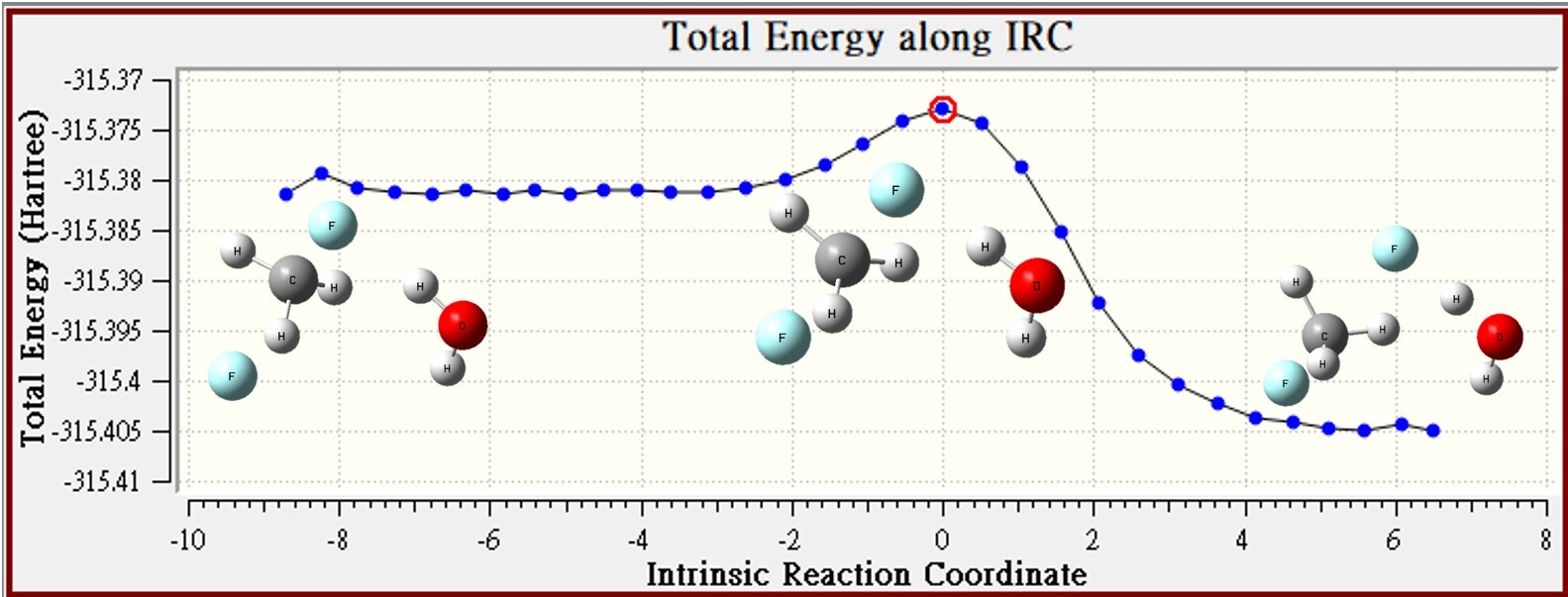
3.Continuum Model ( PCM,sovlent=Water )
Unit for barrier height: kcal/mol ; for other numbers: hartrees
| gas | CH3F | F- | reactant | TS | barrier height |
| Energy | -139.41718 | -99.665948 | -239.08313 | -239.08651 | -2.1 |
| MS | CH3F | F- | reactant | TS | barrier height |
| Energy | -139.41718 | -175.9696 | -315.38678 | -315.37293 | 8.7 |
| CM ( PCM,sovlent=Water ) | CH3F | F- | reactant | TS | barrier height |
| Energy | -139.33783 | -176.08246 | -315.42029 | -315.46832 | -30.1 |