
1.Please use ChatGPT to explore a thematic series of questions within the scope of computational chemistry and follow up with further inquiries about any unfamiliar terms ( except homework )
2.Calculate the structure and frequencies of XHn (X = H~Ne) by using B3LYP theoretical method with 6-31+G(d,p) then compare with the experimental values.
reference:CCCBDB
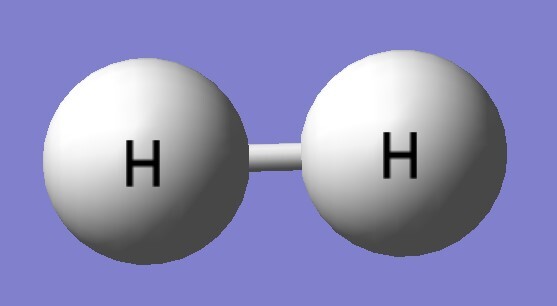
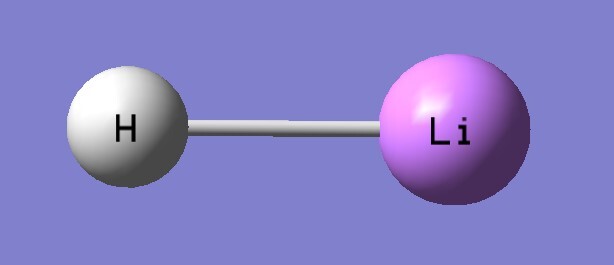
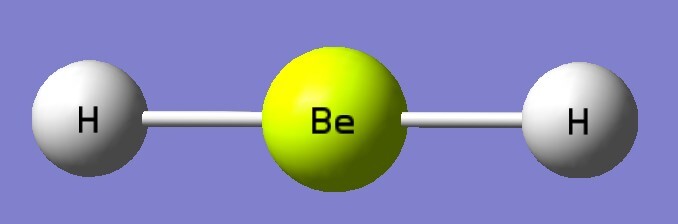
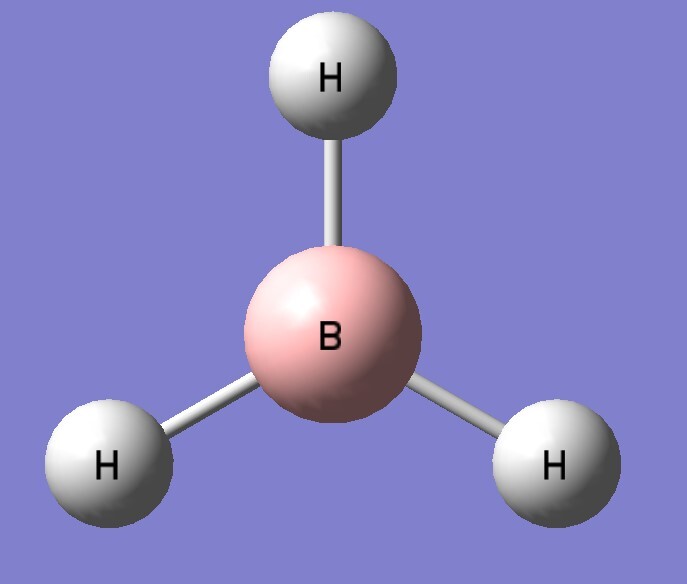
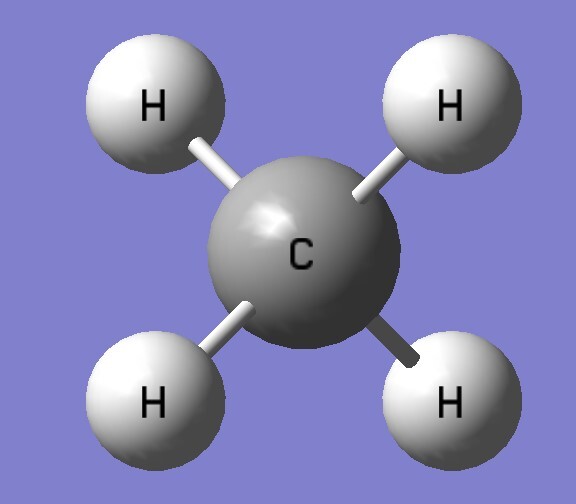
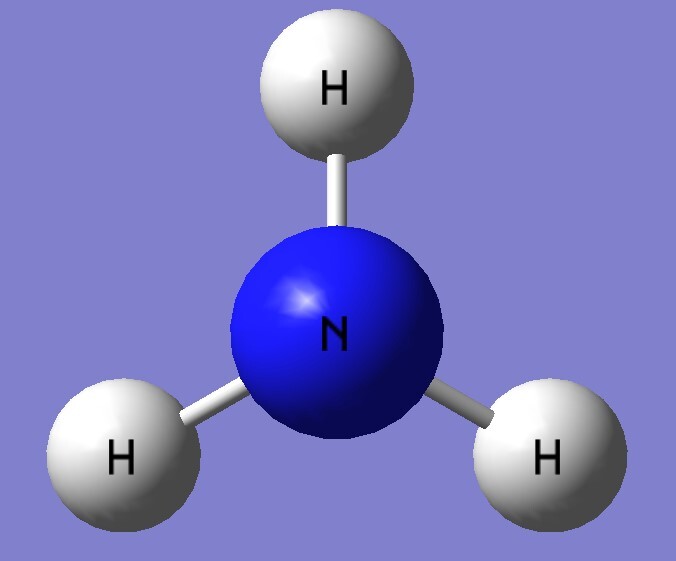
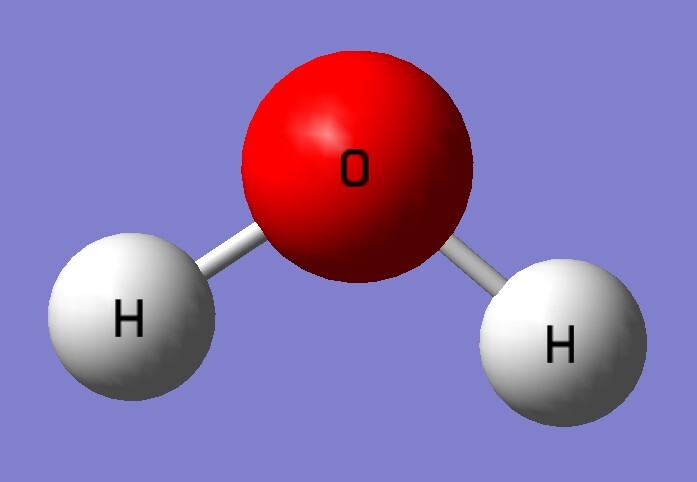
| B3LYP/aptz | bond length(Å) | exp(Å) |
| H2 | 0.743 | 0.741 |
| LiH | 1.614 | 1.595 |
| BeH2 | 1.331 | 1.326 |
| BH3 | 1.193 | 1.19 |
| CH4 | 1.093 | 1.087 |
| NH3 | 1.016 | 1.012 |
| H2O | 0.965 | 0.958 |
| HF | 0.928 | 0.917 |
| B3LYP/aptz | vibration levels(cm-1) | exp(cm-1) |
| H2 | 4465 | 4161 |
| LiH | 1401 | 1405 |
| BeH2 | 735*2, 2038, 2260 | 698, 2159 |
| BH3 | 1157, 1205*2, 2576, 2704*2... | 1148, 1197, 2602 |
| CH4 | 1347*3, 1563*2, 3037, 3150*3 | 1380*3, 1412*2 , 3104*3, 3215 |
| NH3 | 1000, 1673*2,3484,3627*2 | 1139,1765*2,,3464*2,,3534 |
| H2O | 1603, 3809, 3931 | 1595, 3657, 3756 |
| HF | 4069 | 3961 |
| molecule | Error(cm-1) |
| H2 | 304 |
|
LiH |
4 |
| BeH2 | 37, |
| BH3 | 9, 8, |
| CH4 | 33, 151, 178, 46 |
| NH3 | 139, 92, 50, 163 |
| H2O | 8, 152, 175 |
| HF | 108 |
3.Calculate the single point energy of XHn (X = H~Ne) by using CCSD(T) theoretical method with aug-cc-pVTZ ( the structure obtained from the previous homework ), and then use those energy to calculate the standard enthalpy of formation (ΔH°f) then compare with the experimental values.
Hint : CCSD(T)/aug-cc-pVTZ geom=allchk guess=read freq(ReadFC)
| △Hf | CCSD(T) | exp value |
| H2 | 0 | 0 |
| LiH | 26.2 | 33.6 |
| BeH2 | 16.8 | 30 |
| BH3 | -3.8 | 21 |
| CH4 | -56.8 | -17.8 |
| NH3 | -49.4 | -11 |
| H2O | -91.4 | -57.8 |
| HF | 25.2 | -65.3 |
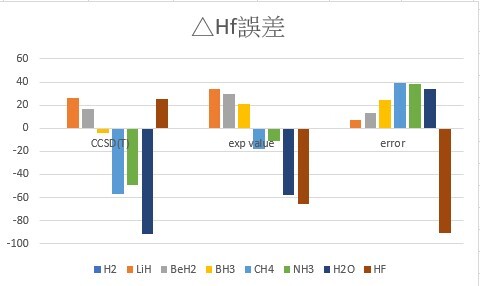
我認為誤差都大到不可接受,